Shafiqul Islam
Abstract

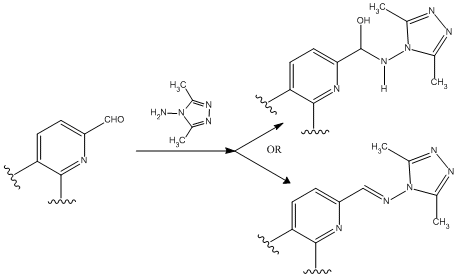
1,2,4-Triazoles and their derivatives have attracted significant attention in several different areas. These nitrogen-rich compounds represent one of the most biologically active classes of the chemical species. This arises from their ability to bind to a variety of enzymes and receptors in biological system via diverse non-covalent interactions. The Schiff bases obtained from 4-amino-3,5-dimethyl-1,2,4-triazole and their metal complexes have shown to inhibit endocytosis and protein tyrosine phosphatases respectively. Recently, the stable hemiaminals coming from the reaction between 4-amino-1,2,4-triazole and substituted benzaldehydes under neutral conditions were obtained. The present paper describes a novel application of 4-amino-3,5-dimethyl-1,2,4-triazole in the preparation of Schiff bases and stable hemiaminals from various pyridyl dialdehyde. The effects of temperature, solvent, structure and concentration of the reagents on the product yields and stability are considered. Our results can contribute to a better understanding of the mechanism of hemiaminal formation from aminotriazoles and pyridylaldehydes.
Publications:
- H-Bonding plays a key role in the formation of stable hemiaminals. Md. Shafiqul Islam, Md. Razzak, Mohammad Karim, Aminul H. Mirza, Tetrahedron Letters, 2017, http://dx.doi.org/10.1016/j.tetlet.2017.01.086
- Schiff Bases as Anti-cancer Drugs: Synthesized from 4-amino-3,5-dimethyl-1,2,4-triazole, Phenathroline and Bipyridine Dicarboxaldehydes. Md. Shafiqul Islam,1 Mohammad R. Karim*1, William Boadi,1 Seyi Falekun,1 Aminul H. Mirza.2 Advances in Biological Chemistry, 2016. pp 180-192, DOI: 10.4236/abc.2016.66016
- A. Azim Jambol1, Malai Haniti S. A. Hamid1*, Aminul H. Mirza1, Md. Shafiqul Islam,2 Mohammad R. Karim2. Some Novel Schiff bases from Pyruvic Acid with Amines containing N & S donor atoms: Synthesis, Spectral Studies and X-Ray Characterization. 1Department of Chemical Sciences, Faculty of Science, Universiti Brunei Darussalam, Bandar Seri Begawan, Brunei Darussalam, 2Department of Chemistry, Tennessee State University, Nashville, USA. Email*: haniti.hamid@ubd.edu.bn. IJOC, 2017, 7, 42-56


